 Rutherford’s earlier model of the atom had also assumed that electrons moved in circular orbits around the nucleus and that the atom was held together by the electrostatic attraction between the positively charged nucleus and the negatively charged electron. Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii. 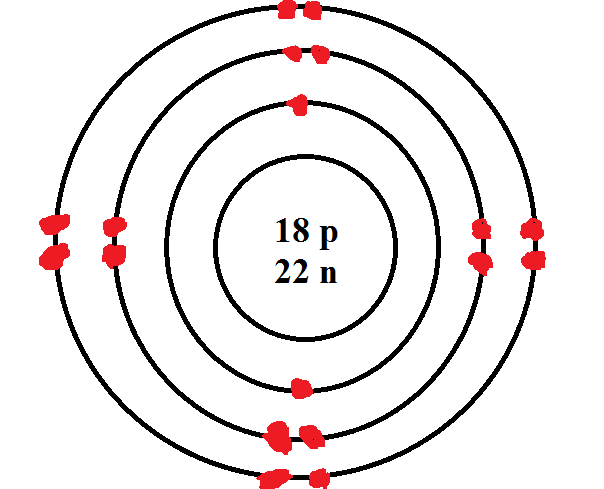
An early model of the atom was developed in 1913 by Danish scientist Niels Bohr (18851962). In 1913, a Danish physicist, Niels Bohr (1885–1962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. How to Draw Atoms (Bohr Model) HighSchoolScience101 27.7K subscribers Subscribe 17K views 1 year ago Chemistry In this lesson I present an overview of: - Different types of atomic models and. Thus, each element, at least when electrically neutral, has a characteristic number of electrons equal to its atomic number. Using the Bohr model, determine the energy, in electron volts, of the photon produced when an electron in a hydrogen atom moves from the orbit with n 5 to the orbit with n 2. The Bohr model shows the atom as a central nucleus containing protons and neutrons, with the electrons in circular electron shells at specific distances from the nucleus, similar to planets orbiting around the sun. 
Where \(n_1\) and \(n_2\) are positive integers, \(n_2 > n_1\), and \( \Re \) the Rydberg constant, has a value of 1.09737 × 10 7 m −1. It is the amount of energy that an electron gains when subjected to a potential of 1 volt 1 eV 1.602 (×) 10 19 J. How to Draw the Bohr-Rutherford Diagram of Carbon chemistNATE 241K subscribers Subscribe 540 Share Save 45K views 3 years ago Carbon has 2 electrons in its first shell and 4 in its second. An early model of the atom was developed in 1913 by the Danish scientist Niels Bohr (18851962). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
